Table of Contents

What is Clinical Data Management?
Definition & Importance
Clinical Data Management (CDM) is the backbone of clinical research. It involves collecting, cleaning, validating, and managing data generated during clinical trials. Imagine running a clinical trial without structured data—it would be like trying to complete a puzzle without knowing what the final picture looks like. CDM ensures that every piece of data is accurate, reliable, and ready for regulatory submission.
In today’s pharmaceutical and biotech industries, CDM plays a critical role in ensuring drug safety and efficacy. Regulatory authorities like CDSCO, FDA, and EMA rely heavily on clean datasets before approving any drug. Without proper clinical data management systems, even a promising drug can fail approval due to poor data quality.
Role in Clinical Trials
CDM professionals act as a bridge between clinical research teams and data analytics teams. They design Case Report Forms (CRFs), manage databases, and perform data cleaning processes. Tools like clinical data management software (e.g., Medidata Rave, Oracle Clinical) are commonly used to ensure data integrity.
They also work closely with biostatisticians and clinical research associates. Their role ensures that trial results are accurate and reproducible. Without CDM, clinical trials would be chaotic, unreliable, and scientifically invalid.

Why Choose Clinical Data Management as a Career?
Industry Demand in India
India has become a global hub for clinical research outsourcing. Many multinational CROs (Contract Research Organizations) are shifting operations to India due to cost efficiency and skilled workforce availability. This has created massive demand for clinical data management jobs.
According to recent industry data, there are hundreds of active job openings in CDM across India. The field is growing at around 12–15% annually, making it one of the fastest-growing healthcare careers.
Growth Opportunities
One of the best things about CDM is career progression. You can start as a Clinical Data Associate and move up to roles like Clinical Data Manager or even Head of Data Management. The growth is not just vertical—you can also switch to related roles like biostatistics, clinical programming, or project management.

Clinical Data Management Salary in India (2026 Updated)
Fresher Salary
If you’re just starting, you might wonder—“Is CDM worth it financially?” The answer is yes, but with patience. According to recent data, freshers earn around ₹2.5–4.5 LPA, with an average of ₹3.5 LPA .
Experienced Salary
With experience, salaries grow significantly. Mid-level professionals earn around ₹5–10 LPA, while senior professionals can earn ₹12–25+ LPA
| Experience Level | Salary Range (India) |
|---|---|
| Fresher | ₹2.5–4.5 LPA |
| 3–5 Years | ₹5–10 LPA |
| 8+ Years | ₹15–25+ LPA |
Types of Clinical Data Management Course
Free Courses
Free courses are a great starting point. They help you understand basics like GCP, CRFs, and data validation without spending money. Many platforms offer free clinical data management courses with certificates.
Paid Courses
Paid courses offer structured learning, mentorship, and placement support. They often include real-world projects and tools training.

Best Free Clinical Data Management Course
| Course Name | Platform | Duration | Certification |
| Data Management for Clinical Research | Coursera (Vanderbilt University) | ~2–6 weeks | Yes (Paid certificate option) |
| Clinical Data Management Course | Elevify | Flexible (4–360 hrs) | Yes (Free Certificate) |
| Clinical Data Management Course | Elevify (Alt Version) | Flexible | Yes |
| Intro Clinical Data Management (YouTube Series) | YouTube | Self-paced | Certificate via provider |
Platforms & Certifications
Some popular platforms offering free clinical data management course online include:
- Coursera (audit mode)
- FutureLearn
- Alison
- WHO Open Learning
These platforms provide free online clinical courses in India with certificates, helping beginners gain foundational knowledge.
Benefits of Free Courses
Free courses are like test-driving a car before buying it. They help you explore the field without commitment. However, they may lack practical exposure and industry tools training.

Best Paid Clinical Data Management Course
| Course Name | Platform | Fees | Features |
| Certificate Course in Clinical Data Management | Udemy | ₹499–₹3,499 (varies) | Basics + CRF + CDM workflow |
| Clinical Data Management (CDM) Online Course | Udemy | ₹499–₹3,999 | Practical modules + regulatory training |
| Certified Clinical Data Management Course | ProRelix Education | ₹15,000–₹40,000 | Placement support + industry exposure |
| Diploma in Clinical Data Management (Oracle Clinical) | ClinSkill | ~$799 (~₹65,000) | Hands-on Oracle Clinical software |
Online Certification Programs
One example is a structured CDM course priced around ₹2999 after discount . These courses often include:
- Placement assistance
- Real-world assignments
- Industry-relevant tools
Key Insights:
- Paid courses include hands-on training, real projects, and placement guidance
- Advanced diploma programs include Oracle Clinical software training, which is highly demanded in CROs
- Udemy courses are affordable and good for quick skill building, but less industry exposure
How to Choose the Right Course (Quick Guide)
- 👉 Beginner? Start with Coursera or Elevify (Free + certification)
- 👉 Want job-ready skills? Go for ProRelix or ClinSkill
- 👉 Low budget? Udemy is best
- 👉 Target MNCs (IQVIA, TCS)? Choose courses with Oracle Clinical / EDC tools
Pro Tip (From PharmD Perspective)
If you’re a Pharm D or life science student, don’t just collect certificates. Focus on:
- CRF design practice
- EDC tools (REDCap, Oracle Clinical)
- Mock datasets
Because recruiters in clinical data management jobs care more about practical exposure than just certificates.

Clinical Data Management Course Syllabus
Core Subjects
A standard clinical data management syllabus includes:
- Introduction to clinical trials
- CRF design
- Data validation
- Query management
- Database locking
Tools & Software Covered
You’ll learn tools like:
- Medidata Rave
- Oracle Clinical
- SAS basics
These tools are essential for clinical data management software operations.
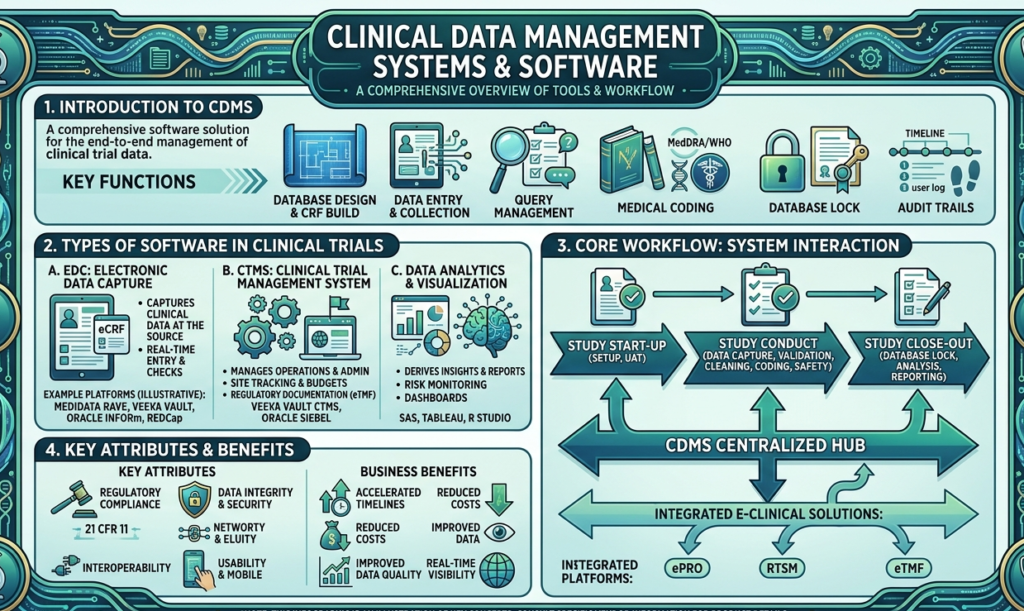
Clinical Data Management Systems & Software
EDC Systems
Electronic Data Capture (EDC) systems are the backbone of CDM. They allow real-time data entry and validation.
Data Validation Tools
Validation tools ensure data accuracy. They flag inconsistencies and missing values, making the dataset reliable.
Clinical Data Management Jobs & Career Roles
Entry-Level Roles
- Clinical Data Associate
- Data Coordinator
- Data Entry Specialist
Advanced Roles
- Clinical Data Manager
- Lead Data Manager
- Data Standards Specialist
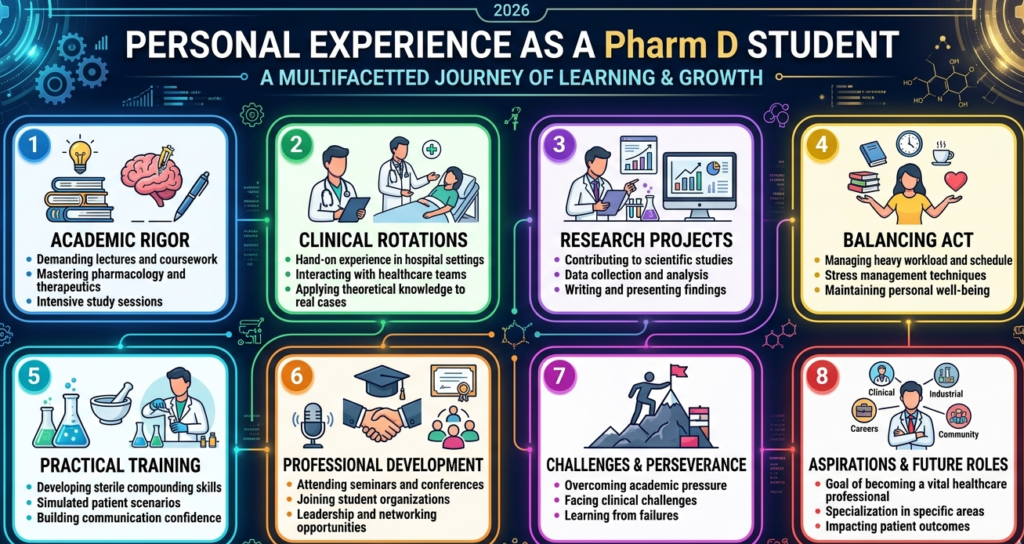
Personal Experience as a Pharm D Student
During my Pharm D journey, I initially focused on clinical practice and patient care. But when I was introduced to clinical research, especially CDM, it felt like discovering a hidden career path. I realized that not everyone needs to be in a hospital setting to make an impact.
I remember working on a mock clinical trial dataset during training. Cleaning messy data and identifying errors felt like solving a detective case. It was challenging but incredibly satisfying. That experience made me understand the importance of accurate data in drug approval.

Case Study Example in Clinical Data Management
Let’s consider a clinical trial for a diabetes drug. During data analysis, inconsistencies were found in patient glucose readings. The CDM team identified that some data entries were incorrect due to manual errors.
After cleaning the data and validating entries, the results changed significantly. The drug’s efficacy became clearer, leading to successful regulatory approval. This shows how CDM directly impacts drug success.
CRA vs CDM – Which is Better?
This is a common question. CRA (Clinical Research Associate) involves fieldwork and site monitoring, while CDM is more desk-based and data-focused.
If you enjoy traveling and interacting with sites, CRA is better. If you prefer data analysis and structured work, CDM is ideal.

Clinical Data Management Companies Hiring in India
Top companies include:
- IQVIA
- Cognizant
- TCS
- Parexel
These companies offer strong career growth and global exposure.
Steps to Start Career in CDM
- Learn basics of clinical research
- Take a clinical data management course online
- Get GCP certification
- Apply for internships
Advantages & Challenges of CDM
CDM offers stability, growth, and global opportunities. However, it can be repetitive and requires high attention to detail.
Future Scope of Clinical Data Management
With AI and automation, CDM is evolving rapidly. However, human expertise is still essential for decision-making and validation.

Conclusion
Clinical Data Management is a powerful career option for pharmacy and life science students. With the right course and skills, you can build a stable and high-paying career. Whether you choose free or paid courses, the key is consistent learning and practical exposure.

FAQs
1. What is the salary of CDM in India?
Freshers earn ₹2.5–4.5 LPA, while experienced professionals can earn ₹15–25+ LPA .
2. What is clinical data management?
It is the process of collecting, cleaning, and managing clinical trial data.
3. What is the salary of Clinical Data Manager in TCS?
Typically ranges between ₹2.8–8 LPA depending on experience .
4. Which is better CRA or CDM?
CRA involves fieldwork, while CDM is data-focused. Choose based on your interest.
5. What are the three phases of CDM?
Data collection, data cleaning, and database locking.

