Table of Contents
If you are a pharmacy, life sciences, or healthcare student staring at two career paths—clinical research and pharmacovigilance—you are not alone. This is one of the most common and honestly one of the most confusing career questions in the pharma world today. One path sounds dynamic and trial-driven. The other sounds stable, regulatory, and deeply tied to drug safety. Both are growing, both are respected, and both can lead to rewarding careers. So naturally, the real question becomes: which is better for you?
That answer is not as simple as “this one pays more” or “that one has more jobs.” In 2025 and 2026, the healthcare industry is changing quickly. Risk-based trials, decentralized studies, AI-assisted safety monitoring, remote data review, and stricter compliance systems are reshaping how companies hire and train talent in both fields. The updated ICH E6(R3) Good Clinical Practice framework, finalized in 2025, is a major reason clinical operations roles are evolving. At the same time, pharmacovigilance is being transformed by automation, signal detection tools, and AI-assisted case workflows rather than disappearing.
So if you have ever searched “pharmacovigilance and clinical research which is better,” “pharmacovigilance and clinical research courses“,“pharmacovigilance vs clinical research salary,” “clinical research coordinator jobs,” or even “entry level pharmacovigilance salary,” this guide is built for you. It is practical, India-focused, beginner-friendly, and honest about what each career really looks like behind the glossy course ads and social media claims. By the end, you will not just understand the difference—you will know where your personality, skills, and long-term goals fit best.
What Is Clinical Research and Pharmacovigilance?
Before comparing both fields, you need to understand what they actually do in the real world—not just what course brochures claim. A lot of students hear these terms and imagine both are part of “pharma jobs,” which is technically true, but that is like saying surgery and radiology are the same because both are in hospitals. They work toward a common healthcare goal, yet their daily responsibilities, tools, work environments, and long-term growth can feel very different.
Build your skills with pharmacovigilance and clinical research courses.
What Is Clinical Research?
If you have ever asked “what is clinical research”, think of it as the science of testing whether a drug, vaccine, device, or treatment actually works in humans—and whether it works safely enough to be approved and used. Clinical research is the engine behind clinical trials, where new interventions are studied under controlled conditions. This field includes protocol development, site coordination, patient recruitment, informed consent, trial documentation, data collection, ethics approvals, compliance, and sponsor communication. In simple words, clinical research is where medicine gets tested before it reaches the market.
This field has become even more important because modern clinical trials are no longer only hospital-based and paper-heavy. The final ICH E6(R3) guidance has pushed the industry toward risk-based quality management, data governance, participant protection, and more flexible trial conduct, which means companies now want professionals who understand both operations and compliance. That is why roles such as clinical research coordinator, clinical research assistant, and clinical research associate continue to attract fresh graduates and postgraduates.
Build your skills with pharmacovigilance and clinical research courses.
What Is Pharmacovigilance and Why Is It Important?
Now let’s answer the equally important question: what is pharmacovigilance? Pharmacovigilance—often shortened to PV—is the science and activity of detecting, assessing, understanding, and preventing adverse effects or any other drug-related problems. If clinical research helps a medicine get to the market, pharmacovigilance watches what happens after and during that journey, especially when real patients start using the product at scale. In other words, if clinical research is the launchpad, pharmacovigilance is the long-term surveillance radar.
This matters because even well-studied medicines can behave differently in larger populations, special patient groups, or when used alongside other drugs. Pharmacovigilance teams collect adverse event reports, process cases, evaluate causality, support aggregate safety reports, and help identify safety signals before they become bigger public health issues. Research and regulatory literature in 2025–2026 shows that pharmacovigilance is also becoming more data-driven, with AI and Bayesian signal detection methods improving how safety teams detect patterns in large datasets. So yes, PV is not just “data entry.” Done well, it protects patients and shapes real regulatory decisions.
Build your skills with pharmacovigilance and clinical research courses.

Clinical Research vs Pharmacovigilance: The Core Difference
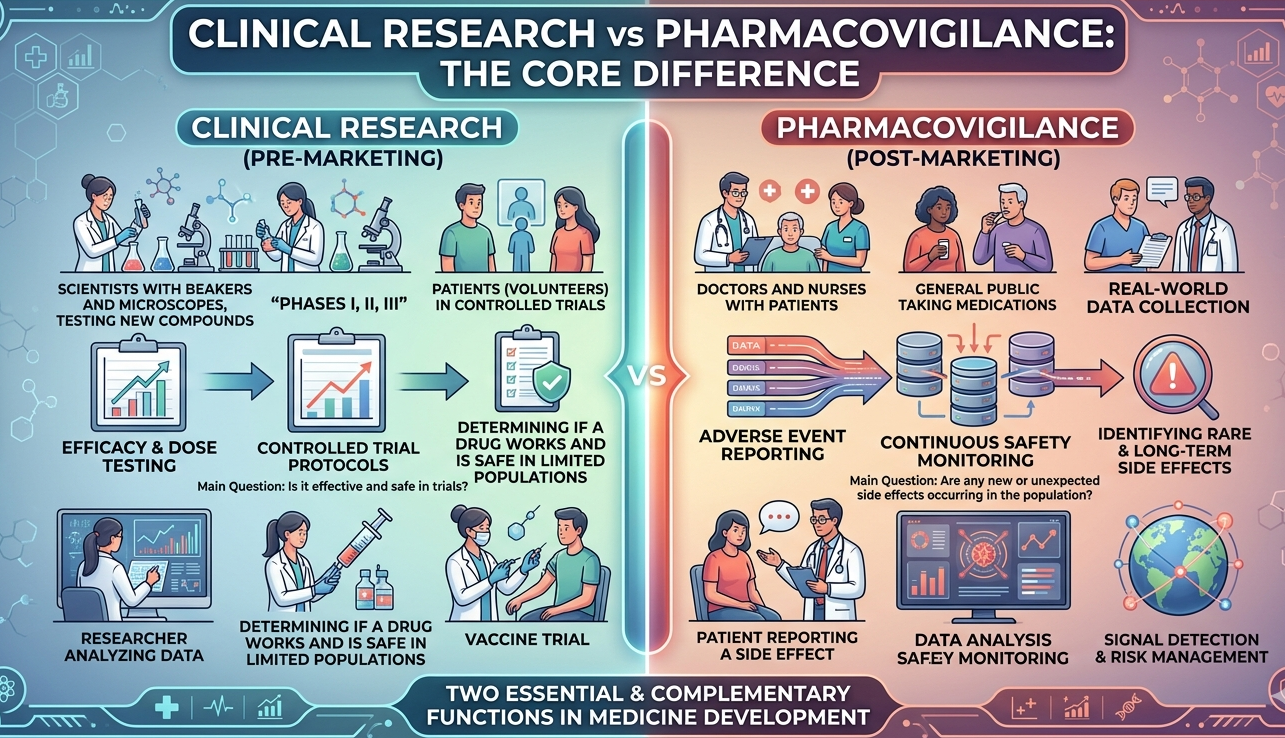
This is the section most students jump to first, and rightly so. If both careers belong to pharma and healthcare, then what is the real difference? The easiest way to understand it is this: clinical research is focused on generating evidence, while pharmacovigilance is focused on monitoring safety. One asks, “Does this treatment work under trial conditions?” The other asks, “What safety issues are emerging as this product is used?”
Build your skills with pharmacovigilance and clinical research courses
Where They Overlap
Both fields are deeply regulated, documentation-heavy, and quality-sensitive. Both require strong scientific understanding, attention to detail, and comfort with SOPs, timelines, and compliance frameworks. In both careers, a small mistake can have serious consequences—whether it is a protocol deviation in a clinical trial or an incorrectly coded adverse event in a safety database. Both also involve collaboration with sponsors, CROs, hospitals, physicians, ethics teams, and regulatory professionals. So if you are looking for a structured career in healthcare without necessarily doing direct bedside patient care, both can be excellent options.
There is also a practical overlap in the kind of students these fields attract. PharmD, B.Pharm, M.Pharm, BSc Life Sciences, MSc Clinical Research, Biotechnology, Microbiology, and even MBBS graduates often explore both. In fact, many job seekers search for pharmacovigilance and clinical research courses together because the foundational knowledge—GCP, medical terminology, adverse event reporting, protocol understanding, and regulatory basics—can support both directions.
Build your skills with pharmacovigilance and clinical research courses
Where They Separate
But once you enter the work itself, the difference becomes clearer. In clinical research, your work is often tied to trial operations. You may be coordinating patients, maintaining site files, scheduling visits, ensuring source documentation, communicating with monitors, and making sure the trial runs according to protocol. In pharmacovigilance, your work is more often tied to drug safety workflows such as ICSR processing, MedDRA coding, case triage, aggregate reporting, signal review, literature surveillance, and regulatory submission support.
Here is the blunt truth: clinical research feels more operational and people-facing, while pharmacovigilance feels more analytical, process-driven, and document-intensive. That does not make one better than the other. It simply means your fit depends on whether you prefer the movement and coordination of trial execution or the precision and safety intelligence of post-marketing and ongoing safety surveillance.
Build your skills with pharmacovigilance and clinical research courses

Career Scope in India in 2026
If you are in India, this comparison becomes even more interesting because both fields have strong visibility, but the hiring patterns are not identical. Students often hear that “clinical research has more glamour” while “pharmacovigilance has more backend stability.” That stereotype contains a grain of truth, but it is far from the full picture.
Build your skills with pharmacovigilance and clinical research courses
Growth of Clinical Research Jobs
India continues to remain attractive for clinical operations, site management, medical writing, data management, and CRO-based trial support. As global sponsors push for efficient trial execution and cost-conscious operations, India remains relevant not just for manpower but also for scientific and operational expertise. The updated GCP environment is also pushing organizations to hire people who understand compliance, digital systems, and quality-by-design—not just paperwork. This has created visible demand for roles such as clinical research coordinator jobs, clinical research assistant, clinical research associate, and clinical trials research associate across hospitals, CROs, academic centers, and sponsor organizations.
Build your skills with pharmacovigilance and clinical research courses
Demand for Pharmacovigilance Roles
Pharmacovigilance, meanwhile, remains one of the most accessible and scalable non-clinical pathways for pharmacy and life sciences graduates in India. Large service hubs, global capability centers, and outsourced safety operations have kept PV hiring active, especially in metro hubs such as Pune, Hyderabad, Bengaluru, Mumbai, and Ahmedabad. Even recent role postings show that companies continue to hire PV coordinators and drug safety professionals, particularly where compliance and reporting timelines matter.
The more important point, though, is this: pharmacovigilance is not “dying because of AI.” That fear gets repeated far too often. What is actually happening is task transformation. Routine literature screening, label extraction, and pattern recognition are increasingly being supported by automation and machine learning, but that only raises the value of professionals who can interpret signals, understand medical context, and make sound safety judgments. So the better question is not “Will AI replace pharmacovigilance?” but “Can you build a PV profile that works well with AI-enhanced workflows?” The answer is yes—very much yes.
Build your skills with pharmacovigilance and clinical research courses

Job Roles in Clinical Research
Clinical research has a ladder-like career structure, which is good news for freshers. You can enter through coordination or assistant roles and gradually move into monitoring, project support, trial management, quality, or even medical affairs-adjacent functions. The path is rarely instant, but it is visible.
Clinical Research Coordinator
The clinical research coordinator is often the backbone of a trial site. If the trial is a stage play, the principal investigator may be the star, but the coordinator is the stage manager making sure the lights, script, timing, and cast all stay aligned. A clinical research coordinator may handle participant scheduling, informed consent logistics, source document maintenance, ethics committee communication, protocol adherence, visit tracking, investigational product accountability, and coordination with sponsor monitors.
If you are wondering how to be a clinical research coordinator, start by building comfort with GCP, trial documents, patient interaction, hospital workflow, and documentation discipline. A lot of students underestimate how much organization and communication this role demands. It is not just “helping with research.” It is structured operational responsibility with regulatory consequences.
Build your skills with pharmacovigilance and clinical research courses
Clinical Research Assistant
A clinical research assistant is often the most beginner-friendly entry point into this field. This role usually supports coordinators, site teams, or project teams by helping with filing, data entry, trial master file updates, participant documentation, screening logs, scheduling support, and basic administrative coordination. It may sound junior, but it can be an excellent launchpad because it teaches you the grammar of trial work. Think of it like learning music scales before performing a concerto—you need the basics to grow confidently.
Build your skills with pharmacovigilance and clinical research courses
Clinical Research Associate
The clinical research associate (CRA) is one of the most sought-after roles in the industry because it is closely associated with sponsor and CRO-side trial oversight. A CRA monitors clinical trial sites, checks protocol compliance, reviews source data, ensures documentation quality, verifies informed consent, and flags deviations or risks. This role often requires travel, communication maturity, strong observational skills, and comfort with site oversight. Many students specifically aim for CRA roles because they are seen as more dynamic and often offer stronger long-term growth than purely administrative entry roles.
Build your skills with pharmacovigilance and clinical research courses
Clinical Trials Research Associate
The term clinical trials research associate is often used interchangeably in job searches, though responsibilities can vary by employer. In practice, these roles usually sit somewhere within trial operations, coordination, or monitoring support. If you see this title in job portals, read the JD carefully. Sometimes it is essentially a CRA-track role; other times it is more site-support oriented.
Build your skills with pharmacovigilance and clinical research courses
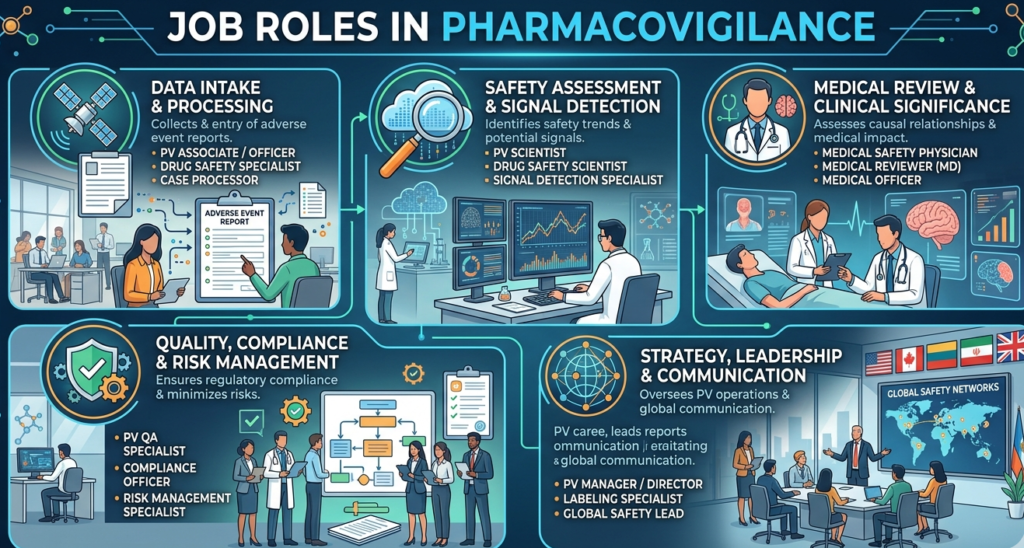
Job Roles in Pharmacovigilance
Pharmacovigilance may look narrower from the outside, but inside the field, it has multiple subtracks. Freshers usually start in case processing or safety operations, but with experience, they can move into signal detection, aggregate reporting, risk management, medical review, quality, or safety science.
Pharmacovigilance Job Profile
A typical pharmacovigilance job profile for entry-level candidates may include case intake, triage, adverse event review, narrative writing, seriousness assessment support, literature review, MedDRA coding, follow-up handling, and database entry using systems such as Argus or similar safety platforms. This is why students who enjoy structured, detail-oriented work often adapt well to PV. It is less about patient scheduling and more about precision, interpretation, and timeline discipline.
Build your skills with pharmacovigilance and clinical research courses
Pharmacovigilance Scientist Job Description
As you move up, the pharmacovigilance scientist job description becomes much more strategic. Higher-level professionals may support product safety strategy, signal management, aggregate reports, periodic safety updates, cross-functional safety review meetings, and interactions with medical reviewers and regulatory teams. Job description databases consistently show responsibilities such as supporting case teams, maintaining product knowledge, collaborating with safety physicians, and planning quality deliverables around surveillance and reporting.
This is where many students get surprised: pharmacovigilance can become very intellectually rich if you stay in it long enough. The field rewards people who can combine medical logic, regulatory thinking, and data interpretation. It may not always feel flashy on day one, but it can evolve into highly meaningful work.
Build your skills with pharmacovigilance and clinical research courses

Skills You Need to Succeed
If you are trying to decide between these two fields, do not only compare salaries and job titles. Compare your natural strengths. That is often the smarter career compass.
Skills for Clinical Research
Clinical research tends to reward people who are organized, responsive, people-friendly, process-aware, and calm under operational pressure. If you are someone who can coordinate moving parts without dropping the ball, this field can suit you very well. You need to be comfortable with patient-facing environments, scheduling, documentation, site communication, and protocol discipline. It also helps if you can talk clearly and professionally because much of this field depends on communication—between investigators, sponsors, ethics committees, and site teams.
A major advantage here is that these are transferable skills. Once you become strong in coordination and trial operations, you can grow into CRA, project associate, trial manager, quality roles, or related sponsor-side positions. Clinical research often rewards those who can combine scientific understanding with real-world execution.
Build your skills with pharmacovigilance and clinical research courses
Skills for Pharmacovigilance
Pharmacovigilance, on the other hand, tends to reward people who are methodical, analytical, medically curious, patient with repetitive detail, and good at spotting inconsistencies. If you are the type who notices tiny errors in forms, enjoys classification logic, and can work through complex information without losing accuracy, PV may fit you better than you expect.
You also need comfort with medical terminology, adverse event language, coding logic, case narratives, and safety timelines. In a way, PV is like being a detective for drug safety. You are not always in the spotlight, but your work helps identify the clues that protect patients. That is a serious responsibility—and for the right person, a deeply satisfying one.
Build your skills with pharmacovigilance and clinical research courses
Salary Comparison
Money should not be the only factor in a career decision, but pretending it does not matter would be unrealistic. Students frequently search “pharmacovigilance vs clinical research salary” because they want a practical answer: which path pays better?
Pharmacovigilance vs Clinical Research Salary
The honest answer is this: at entry level in India, the salary difference is often not dramatic—but the growth pattern can differ depending on role, city, company type, and skill stack. Clinical research roles tied to hospitals or smaller sites may begin modestly, especially for clinical research assistant or clinical research coordinator positions. Sponsor-side or CRO-side roles like CRA can improve faster with experience. Pharmacovigilance entry roles can also begin at moderate levels, especially in outsourced operations, but professionals with niche safety, medical review, or aggregate reporting expertise can eventually command stronger compensation.
Community discussions on Indian pharmacy and medical career forums suggest that salary growth in PV can feel slower in some service-heavy tracks unless you switch strategically or move into higher-value subdomains. Meanwhile, broader non-clinical healthcare hiring posts in India continue to show demand across clinical research, drug safety, regulatory, and medical writing, with compensation varying sharply by qualification and role complexity.
So, entry level pharmacovigilance salary may not always wow freshers at first glance, and neither will all entry-level clinical research roles. The smarter move is to choose the field where you can become really good, because long-term earnings usually follow skill depth, not just job title hype.
To increase your chances build your skills by enrolling with pharmacovigilance and clinical research courses
Quick Comparison Table
| Factor | Clinical Research | Pharmacovigilance |
| Best for | Operational, people-facing professionals | Analytical, safety-focused professionals |
| Freshers’ entry roles | Clinical research assistant, clinical research coordinator | Drug safety associate, PV associate |
| Work style | Trial execution and coordination | Case processing and safety surveillance |
| Growth route | CRA, trial manager, project roles | Safety scientist, aggregate reporting, signal detection |
| Travel chances | Moderate to high in some roles | Usually low to minimal |
| Documentation load | High | Very high |
| AI impact | Workflow modernization | Automation support, not full replacement |
| Salary growth | Strong with CRA/project path | Strong with specialization and switching |

Courses and Certifications
This is where many students make avoidable mistakes. They collect certificates like souvenirs but do not build employable competence. A certificate can help, yes—but only if it is tied to skill, clarity, and interview readiness.
Clinical Research Certification Options
If you want to enter clinical research, useful learning areas include GCP, clinical trial phases, ethics, protocol basics, source documentation, informed consent, trial monitoring, and regulatory frameworks. Searches for clinical research certification, clinical research certificate course, and clinical coordinator certification are common because students want something that adds credibility to their CV. That is sensible—but choose programs that actually teach workflows, not just definitions.
If you are specifically aiming for a site role, prioritize training that helps you understand trial file handling, patient flow, ethics communication, AE reporting basics, and audit readiness. If you want to move toward CRA-type roles later, learn monitoring logic, source verification principles, and sponsor expectations early.
Build your skills with pharmacovigilance and clinical research courses
Pharmacovigilance Courses
For PV, the best learning path usually includes pharmacovigilance basics, ICSR workflow, adverse event terminology, seriousness/expectedness concepts, MedDRA overview, signal detection basics, aggregate reporting awareness, and safety databases. Searches like pharmacovigilance courses, pharmacovigilance online courses with certificate, and pharmacovigilance and clinical research courses are rising because students increasingly want flexible learning options.
The key is to avoid getting seduced by course marketing that promises “100% placement” without teaching actual job readiness. A smart PV course should help you understand not only what a case is, but why it matters, how it is assessed, and where it fits in the safety ecosystem.
Build your skills with pharmacovigilance and clinical research courses
Free Courses with Certificates
Now to the part students love: free learning options. Yes, people are actively searching for free clinical research courses with certificates, free online clinical research courses in India with certificates , pharmacovigilance and clinical research courses, free clinical data management courses with certificates, and pharmacovigilance free online courses with certificate in India. That demand is real. Free courses can be useful, especially at the exploration stage, but they should be treated as entry doors, not career guarantees.
A practical way to use them is this:
- Take a free foundational course to understand the field.
- Build notes and vocabulary from it.
- Add one applied skill layer—such as GCP for clinical research or case processing basics for PV.
- Practice interview questions and job descriptions.
That four-step method is far more effective than stacking ten random certificates and hoping recruiters will be impressed.Here are some pharmacovigilance and clinical research courses.
Top Free Clinical Research and Pharmacovigilance Courses with Certificates
| Field | Course | What You’ll Learn | Who Should Join |
| Clinical Research | Essentials of Clinical Research | Clinical trial basics, ethics, compliance, GCP | Pharmacy, life sciences, and healthcare students |
| Clinical Research | Good Clinical Practice (ICH E6 R3) | GCP principles, compliance, trial conduct | Aspiring CRCs, CRAs, and research assistants |
| Pharmacovigilance | Introduction to Pharmacovigilance | Drug safety, ADRs, compliance basics | Beginners in PV and drug safety |
| Pharmacovigilance | GVP Training Free | Good Pharmacovigilance Practices, safety principles | Pharmacy and pharma students |
| Pharmacovigilance | Pharmacovigilance Course | ICSRs, signal detection, reporting concepts | Freshers exploring PV careers |
Which Is Better for Freshers?
This is the heart of the article, so let’s answer it directly: pharmacovigilance and clinical research—which is better? The truth is, neither is universally “better.” The better field is the one that matches your strengths, patience level, communication style, and long-term tolerance for the work itself.
If you enjoy people coordination, hospital settings, trial operations, documentation flow, and active communication, then clinical research may be the stronger fit. If you like work that feels dynamic, visible, and operational, this path often feels more alive. It can also be especially attractive if you want to eventually move toward CRA, project coordination, monitoring, or broader trial management.
If you prefer structured desk-based work, medical logic, safety analysis, documentation precision, and less site-facing activity, then pharmacovigilance may be a better long-term home. It can be a very smart choice for students who are detail-oriented and comfortable working through regulated workflows with high accuracy.
So who wins for freshers? Here is the most practical answer:
- Choose Clinical Research if you want:
- More operational exposure
- Patient/trial site interaction
- CRA growth potential
- Strong communication-based roles
- Choose Pharmacovigilance if you want:
- Safer desk-based work style
- Strong drug safety domain
- Less travel
- Better fit for analytical personalities
And if you are still confused? Start by reading 10 job descriptions each for clinical research and PV. That exercise alone will often tell you more than any motivational seminar ever could.Build your skills with pharmacovigilance and clinical research courses
People Also Ask
What is clinical research and pharmacovigilance?
Clinical research is the process of studying drugs, devices, or treatments in humans to evaluate safety and effectiveness. Pharmacovigilance is the science of monitoring, detecting, and preventing adverse drug reactions and safety-related issues during and after a product’s use.
Which is better, clinical research or pharmacovigilance?
It depends on your strengths. Clinical research is often better for people who enjoy trial operations, coordination, and communication. Pharmacovigilance is often better for people who prefer structured analytical work, safety monitoring, and documentation-heavy processes.
Will AI replace pharmacovigilance?
No—not fully. AI is already helping with literature screening, case triage, signal support, and automation, but human judgment remains essential for medical interpretation, safety decisions, and regulatory quality. AI will change PV workflows, but it is more likely to reshape jobs than erase them.
Which is better, PV or CDM?
That depends on your interests. PV focuses on drug safety and adverse event monitoring, while CDM (Clinical Data Management) focuses on the quality, structure, and integrity of clinical trial data. CDM may appeal more to data-oriented professionals, while PV is stronger for those interested in safety science.
What are the 4 pillars of pharmacovigilance?
A practical way to understand the four pillars of pharmacovigilance is:
- Detection of adverse events
- Assessment of safety signals
- Understanding the risk-benefit profile
- Prevention/communication of drug-related harm
These pillars help ensure that medicines remain as safe as possible throughout their lifecycle.
Conclusion
If you came here hoping for a one-word answer—clinical research or pharmacovigilance—you probably noticed something important by now: the better choice is not the one with louder marketing. It is the one that aligns with the way you actually work best. Careers are not won by choosing the trendiest label. They are built by choosing the lane where you can stay consistent long enough to become valuable.
Clinical research can be exciting, people-centered, and full of movement. Pharmacovigilance can be stable, intellectually sharp, and deeply meaningful in protecting patients. One is not glamorous and the other boring. That is a shallow myth. Both can become excellent careers if approached strategically. So instead of asking only “Which is better?”, ask the smarter question: “Which one can I genuinely grow in for the next five years?” That question will save you far more time, money, and frustration than any course ad ever will.

FAQs
1. How to be a clinical research coordinator?
Start by learning GCP, clinical trial basics, informed consent, ethics, source documentation, and hospital workflow. Entry roles or internships at hospitals, research centers, or CROs can help you build practical exposure. Strong communication and organization are essential.
2. Is clinical research a good career in India?
Yes, especially for students from pharmacy, life sciences, and healthcare backgrounds. Demand continues in site coordination, monitoring, project support, and sponsor/CRO operations, especially as modern trial systems evolve.
3. Are pharmacovigilance courses worth it?
They can be worth it if they teach actual job skills such as case processing, AE concepts, MedDRA basics, and safety workflows. A certificate alone is not enough—you need interview-ready understanding.
4. What are the top pharmacovigilance companies in India?
Many professionals target global CROs, safety service providers, multinational pharma companies, and capability centers in cities such as Pune, Hyderabad, Bengaluru, and Mumbai. The “best” company depends on your role, team quality, growth path, and training exposure.
5. Can I switch from clinical research to pharmacovigilance or vice versa?
Yes, especially early in your career. The transition is easier when you build common foundations like GCP, AE understanding, regulatory basics, medical terminology, and documentation discipline. The sooner you identify your preferred long-term direction, the smoother the switch becomes.