Table of Contents

🧠 What is ADR Reporting?
ADR reporting refers to the process of documenting and communicating any harmful or unintended effects caused by medications. According to pharmacovigilance definitions, an adverse drug reaction (ADR) is a harmful and unintended response occurring at normal drug doses.
Think of it like a “feedback system” for medicines. Just as apps improve with user reviews, drugs become safer when healthcare professionals report side effects. Without reporting, hidden dangers remain unnoticed, putting thousands of patients at risk.
🔍 Difference Between ADR and Adverse Event
- ADR: Proven relationship with the drug
- Adverse Event: Any medical occurrence (may or may not be drug-related)
Understanding this distinction is crucial when filling an adverse event reporting form or adverse reaction reporting form.
ADR Reporting System in India
India has a structured ADR reporting system in India under the Pharmacovigilance Programme of India (PvPI).
🏥 Pharmacovigilance Programme of India (PvPI)
- Launched in 2010
- Managed by Indian Pharmacopoeia Commission (IPC)
- Collects, analyzes, and monitors drug safety data
Its goal? Ensure that the benefits of medicines outweigh risks.
📊 Growth of ADR Monitoring Centres
Here’s something surprising:
| Year | ADR Monitoring Centres |
|---|---|
| 2010 | 22 |
| 2025 | 1,120+ |
India now has over 1,120 ADR monitoring centres, showing massive growth in drug safety infrastructure.
Yet, underreporting remains a serious issue.
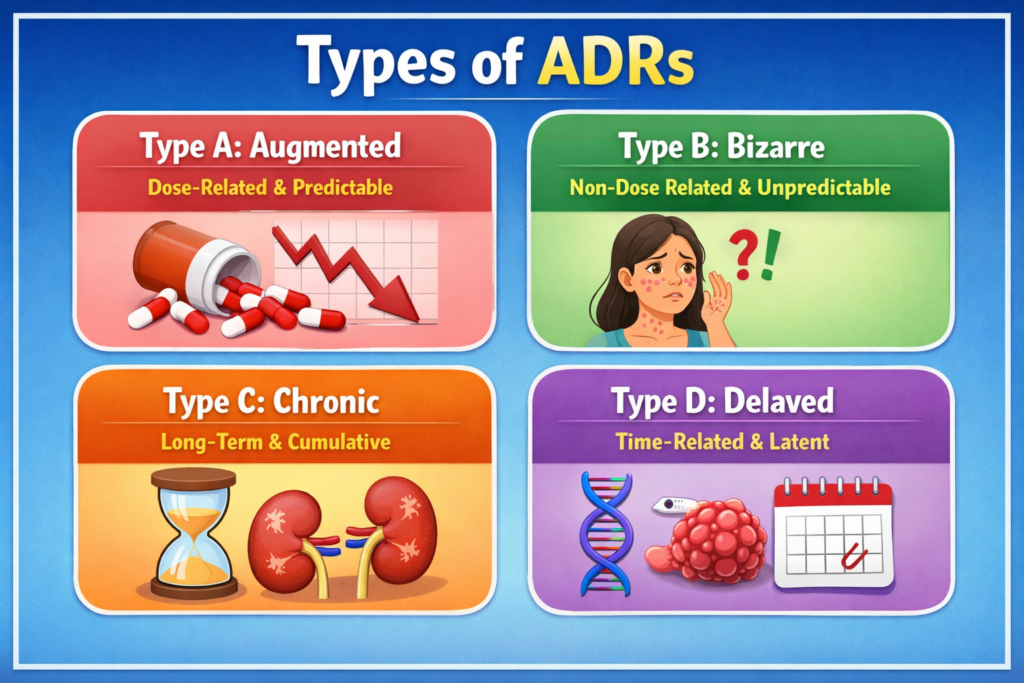
⚠️ Types of ADRs
📌 4 Types of ADRs (Common Classification)
- Type A (Augmented) – Dose-dependent (e.g., bleeding with warfarin)
- Type B (Bizarre) – Unpredictable (e.g., anaphylaxis)
- Type C (Chronic) – Long-term use effects
- Type D (Delayed) – Appears after time
👉 Type B ADRs are unpredictable, making them the most dangerous.
📌 6 Types of ADR Classification
- Type A – Augmented
- Type B – Bizarre
- Type C – Chronic
- Type D – Delayed
- Type E – End-of-use
- Type F – Failure of therapy
👩⚕️ Who Can Report ADR in India?
This is one of the most misunderstood areas.
✔ Doctors
✔ Pharmacists
✔ Nurses
✔ Dentists
✔ Patients / Consumers
Yes, anyone can report ADRs, not just healthcare professionals.
📍 Where to Report ADR?
You can report ADR through:
- Nearest ADR Monitoring Centre (AMC)
- PvPI helpline: 1800-180-3024
- Email submission
- Mobile apps
- Hospitals
🏥 ADR Reporting Centre in India / ADR reporting system in India
These centres are located in:
- Medical colleges
- Government hospitals
- Private institutions

📝 Step-by-Step Guide to ADR Reporting System in India
✅ Step 1: Identify the ADR
Look for:
- Unexpected symptoms
- Drug interactions
- Allergic reactions
✅ Step 2: Collect Patient Information
Include:
- Age, gender
- Medical history
- Drug details
✅ Step 3: Fill ADR Reporting Form
Use:
- ADR report form
- Adverse drug reaction reporting form
- ADR reporting form for consumer
✅ Step 4: Submit the Form
Submit via:
- Hospital AMC
- Online
- Helpline
✅ Step 5: Follow-Up
Provide additional data if requested.

📄 ADR Reporting Forms
🧾 PvPI ADR Reporting Form
- Official Indian form
- Used by healthcare professionals
- Includes drug, reaction, and patient data
🧾 ADR Reporting Form for Consumer
- Simplified format
- Available in multiple languages
🧾 FDA Adverse Event Reporting Form
- Used globally (USA)
- Known as MedWatch Form
💻 Software Used in ADR Reporting System in India
- VigiFlow – Used by PvPI for data collection
- VigiBase – WHO global database
These tools help detect safety signals worldwide.

👨⚕️ Personal Experience (Pharm D Intern)
During my Pharm D internship, I encountered a patient who developed a severe rash after taking amoxicillin. Initially, it seemed like a simple allergy, but documenting it changed everything.
I filled out an ADR reporting form, noting:
- Drug history
- Onset timing
- Severity
The case was forwarded to the ADR monitoring centre. Weeks later, I realized that even one report contributes to national safety data.
That moment made me understand:
👉 ADR reporting is not paperwork—it’s patient protection.
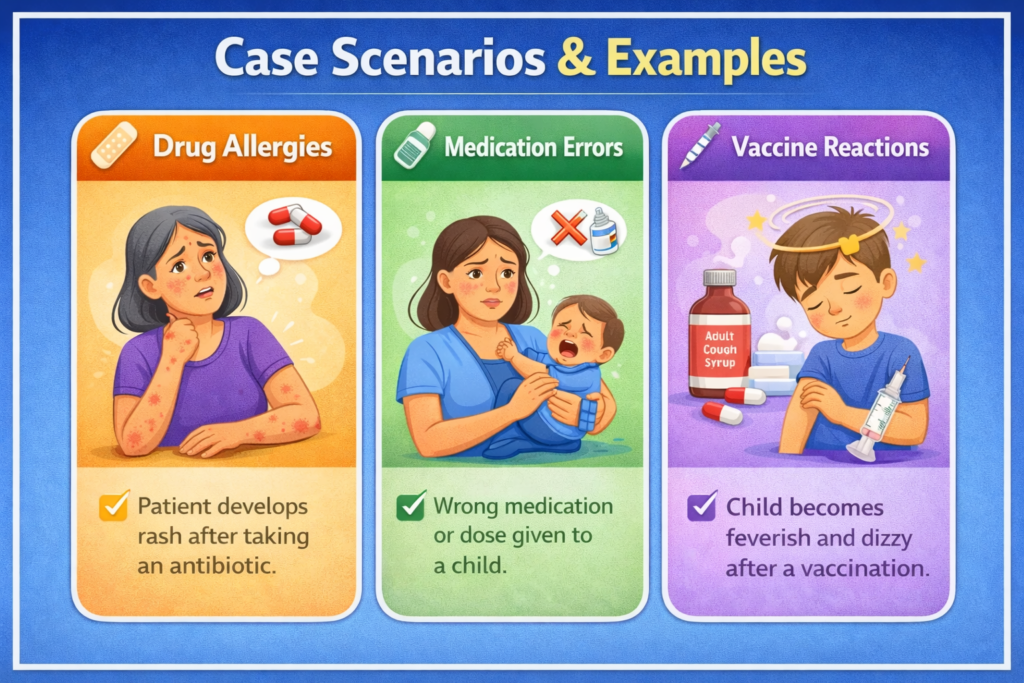
📚 Case Scenarios & Examples
🧪 Case 1: Antibiotic Reaction
A patient develops diarrhea after antibiotics.
➡ Report as Type A ADR
💉 Case 2: Vaccine Reaction
Mild fever post-vaccination
➡ Usually expected ADR
🚨 Case 3: Anaphylaxis
Sudden breathing difficulty after drug intake
➡ Type B ADR (unpredictable)
🎯 Importance of ADR Reporting
- Improves drug safety
- Detects rare side effects
- Prevents future harm
- Supports regulatory decisions
India contributes significantly to global safety databases, but underreporting is still a major gap.
⚡ Challenges in ADR Reporting
- Lack of awareness
- Fear of legal consequences
- Time constraints
- Poor documentation

📜 Guidelines for ADR Reporting in India
- Report all suspected ADRs
- Report even if unsure
- No legal liability
- Maintain patient confidentiality

🧾 People Also Ask (FAQs)
❓ What is the ADR reporting system in India?
It is a national system under PvPI that collects and analyzes adverse drug reactions to improve drug safety.
❓ Who can report an ADR in India?
Anyone including doctors, pharmacists, nurses, and patients can report ADRs.
❓ What software is used in ADR reporting?
VigiFlow and VigiBase are commonly used.
❓ What is ADR reporting?
It is the process of documenting adverse drug reactions.
❓ What are the 4 types of ADR?
Type A, B, C, and D.
❓ What are the 6 types of ADRs?
Type A to Type F classification.
❓ How are ADRs reported?
Through forms, mobile apps, helpline, or ADR centres.
❓ Who is most at risk for ADRs?
Elderly, children, and patients on multiple drugs.
❓ Who should report ADRs?
All healthcare professionals and consumers.
❓ Why do companies issue ADRs?
To monitor drug safety and comply with regulations.
❓ Which ADR is unpredictable?
Type B (Bizarre) ADR.

🏁 Conclusion
The ADR reporting system in India has evolved into a robust network with over 1,100 monitoring centres, yet its success depends on one simple action—reporting. Every report, no matter how small, contributes to a larger safety net protecting millions of patients. As future pharmacists, doctors, or even informed patients, embracing ADR reporting is not optional—it’s a responsibility.
